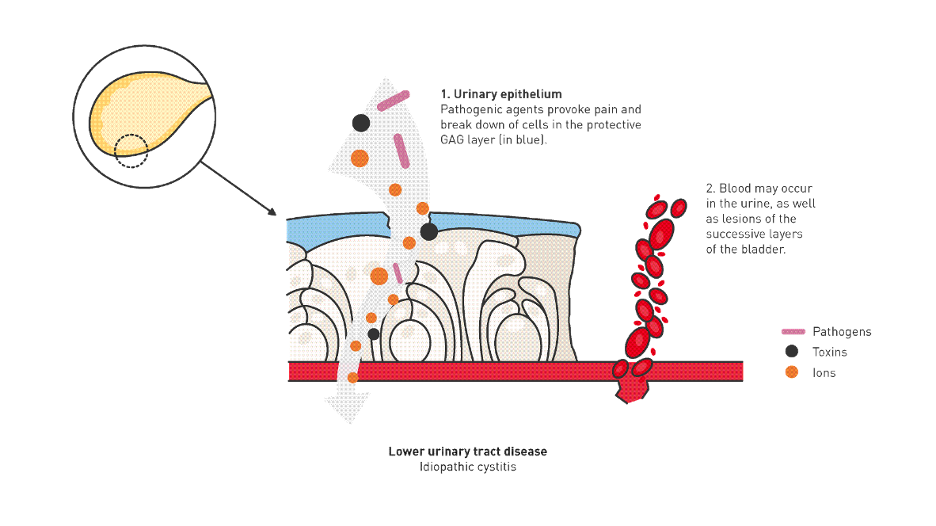
Feline lower urinary tract disease (FLUTD) is a broad term describing disorders that affect the bladder or urethra. It is a relatively frequent reason for veterinary visits, occurring in about 1.5% of cats seen in primary care [1]. FLUTD encompasses a variety of conditions such as urolithiasis, urinary infection, anatomical abnormalities, behavioural issues, and neoplasia. However, more than half of affected cats have no identifiable underlying cause, and these cases are classified as Feline Idiopathic Cystitis (FIC) [2].
Because diagnosing and managing FIC can be challenging, inappropriate urination remains a major contributor to feline relinquishment to shelters [3].
Clinical Signs and Presentation
Cats with FIC typically show the same clinical signs seen in other lower urinary tract disorders, including:
- Difficulty or discomfort during urination
- Increased frequency of urination
- Urinating outside the litter box
- Blood in the urine

Since these signs overlap with many other conditions, FIC is a diagnosis of exclusion, which includes ruling out behavioural causes.
Current understanding suggests that FIC represents a broader stress-related syndrome, involving chronic activation of central nervous system threat pathways and affecting multiple organ systems [4].
Epidemiology and Signalment
There is no universal breed predisposition, although patterns vary by region. Studies have shown:
- A higher risk in Persian cats
- A lower risk in Siamese cats [5, 6]
Other factors strongly associated with FIC include:
- Obesity
- Low activity levels
- Nervous or anxious temperament
- Frequent dietary changes [7, 8]
The condition most commonly affects male cats between 4 and 7 years of age [9].
Environmental influences also play a significant role. Cats with FIC are more likely to:
- Live in apartments
- Live in multicat households
- Use non-clumping litter
- Lack access to elevated resting areas [10]
These findings support the concept that FIC develops when susceptible cats are exposed to stressful or suboptimal environments.
Resolution and Recurrence
Clinical signs of FIC typically resolve within seven days, regardless of treatment [9]. However:
- Up to 65% of cats experience recurrent episodes within a year [8]
- Approximately 15% develop chronic FIC, with signs persisting for weeks or months
Because signs often improve quickly, any treatment may appear effective. This underscores the importance of evidence-based decision-making, especially since administering medication can itself be stressful for cats.
Treatment Strategies
A detailed history—including environmental factors, household dynamics, and daily routines—is essential for identifying risk factors and guiding management.
Stress Reduction
Multimodal Environmental Modification (MEMO) is one of the most effective long-term strategies for reducing FIC episodes. MEMO focuses on decreasing activation of the feline stress response by improving:
- Social interactions
- Resource availability
- Environmental enrichment
In a key study, MEMO resulted in a highly significant reduction in clinical episodes (p < 0.0001), with 75% of cats experiencing no episodes during a 10-month follow-up period [11].
Recommended environmental adjustments include:
- Providing multiple litter boxes with clumping litter in quiet areas
- Offering several water sources and feeding stations
- Ensuring access to scratching posts, toys, and resting areas
- Increasing vertical space and hiding spots
- Following the “one per cat plus one” rule for all resources
- Using belled collars on aggressive cats to warn more timid individuals
Diet
Dietary factors play an important role in FIC management. Considerations include:
- Nutrient composition
- Moisture content
- Feeding method
- Consistency of diet
Current recommendations favour wet food over dry, because the higher moisture content promotes urine dilution, reduces the concentration of irritants, lowers the risk of crystal formation, and encourages more frequent voiding of urine[12, 2, 13].

Therapeutic diets
- Royal Canin Urinary S/O
(Wet and dry options)- Encourages increased water intake
- Promotes diuresis
- Contains EPA and DHA, which help modulate inflammatory mediator production [15]
- Royal Canin Calm
(Dry format only)- Contains alpha-casozepine and L-tryptophan
- Supports stress reduction [16]
Alpha-casozepine is a milk-derived protein with calming properties, while L-tryptophan is a precursor to serotonin, a neurotransmitter associated with mood regulation.
Feline Facial Pheromone (FFP)
Although research is ongoing, synthetic feline facial pheromones have demonstrated benefits in managing anxiety-related behaviours. They offer:
- Non-invasive administration
- Household-wide effects
- Support for both dominant and timid cats
Given the multifactorial nature of FIC, pheromones can be a useful component of a multimodal treatment plan.
Postbiotics and L-tryptophan+alpha casozepine
Serenicare (Protexin) is a nutritional supplement designed to support emotional balance in cats experiencing stress-related conditions such as FIC.
The combination of α‑casozepine and L‑tryptophan -both incorporated into the Royal Canin Calm diet- and postbiotics (HT Bifidobacterium longumandHT Lactobacillus Rhamnosus) targets key neurochemical pathways involved in the feline stress response. Incorporating postbiotics alongside α‑casozepine and L‑tryptophan significantly enhances mood modulation and promotes relaxation.
L‑tryptophan supports serotonin production in the brain, serotonin is a hormone and neurotransmitter that helps in regulating the mood, reduces anxiety, and improves emotional stability.
α‑Casozepine enhances GABA‑A receptor activity, strengthening the brain’s natural calming pathways and promoting relaxation without sedation.The inclusion of postbiotics further supports gut–brain axis modulation, enhancing resilience to stress. With a palatable formulation that minimizes handling stress.
Glycosaminoglycans (GAGs)
A compromised GAG layer may contribute to FIC pathogenesis. Supplements such as Cystophan, which contains N-acetyl-D-glucosamine, hyaluronic acid, and L-tryptophan, aim to support bladder lining integrity.
Although clinical evidence for oral or injectable GAGs in reducing recurrence of feline idiopathic cystitis (FIC) remains limited [18,19], several studies have demonstrated that GAG supplementation—particularly N‑acetyl‑D‑glucosamine—does increase detectable GAG concentrations in plasma and urine.
Reports show that NAG supplementation increased both plasma and urinary GAG levels, with FIC cats exhibiting significantly lower urinary GAGs at baseline compared to healthy controls [20,21].
However, no studies have confirmed whether these supplements directly restore or thicken the urothelial GAG layer, and this limitation is largely due to ethical constraints: bladder biopsies and post‑mortem examinations cannot be performed solely for research purposes in client‑owned animals. As a result, while biochemical evidence supports increased GAG availability, structural confirmation of urothelial repair remains ethically and practically unattainable.
Conclusion
FIC is a challenging and often distressing condition for both cats and their owners. Because multiple factors contribute to its development, management should be tailored to each individual case. Environmental modification should always be discussed, taking into account the household’s specific circumstances. When combined with dietary strategies, supplements, and pheromone therapy, MEMO can significantly improve outcomes and enhance quality of life.
References
- Lund, E.M., et al., Health status and population characteristics of dogs and cats examined at private veterinary practices in the United States. J Am Vet Med Assoc, 1999. 214(9): p. 1336-41.
- Buffington, C.A., et al., Clinical evaluation of cats with nonobstructive urinary tract diseases. J Am Vet Med Assoc, 1997. 210(1): p. 46-50.
- Patronek, G.J., et al., Risk factors for relinquishment of cats to an animal shelter. J Am Vet Med Assoc, 1996. 209(3): p. 582-8.
- Westropp, J.L., M. Delgado, and C.A.T. Buffington, Chronic Lower Urinary Tract Signs in Cats: Current Understanding of Pathophysiology and Management. Vet Clin North Am Small Anim Pract, 2019. 49(2): p. 187-209.
- Lekcharoensuk, C., C.A. Osborne, and J.P. Lulich, Epidemiologic study of risk factors for lower urinary tract diseases in cats. J Am Vet Med Assoc, 2001. 218(9): p. 1429-35.
- Egenvall, A., et al., Morbidity of insured Swedish cats during 1999-2006 by age, breed, sex, and diagnosis. J Feline Med Surg, 2010. 12(12): p. 948-59.
- Lund, H.S., et al., Risk factors for idiopathic cystitis in Norwegian cats: a matched case-control study. J Feline Med Surg, 2016. 18(6): p. 483-91.
- Defauw, P.A., et al., Risk factors and clinical presentation of cats with feline idiopathic cystitis. J Feline Med Surg, 2011. 13(12): p. 967-75.
- Forrester, S.D. and T.L. Towell, Feline idiopathic cystitis. Vet Clin North Am Small Anim Pract, 2015. 45(4): p. 783-806.
- Kim, Y., et al., Epidemiological study of feline idiopathic cystitis in Seoul, South Korea. J Feline Med Surg, 2018. 20(10): p. 913-921.
- Buffington, C.A., et al., Clinical evaluation of multimodal environmental modification (MEMO) in the management of cats with idiopathic cystitis. J Feline Med Surg, 2006. 8(4): p. 261-8.
- Markwell, P.J., et al., Clinical evaluation of commercially available urinary acidification diets in the management of idiopathic cystitis in cats. J Am Vet Med Assoc, 1999. 214(3): p. 361-5.
- Gunn-Moore, D.A. and C.M. Shenoy, Oral glucosamine and the management of feline idiopathic cystitis. J Feline Med Surg, 2004. 6(4): p. 219-25.
- Naarden, B. and R.J. Corbee, The effect of a therapeutic urinary stress diet on the short-term recurrence of feline idiopathic cystitis. Vet Med Sci, 2020. 6(1): p. 32-38.
- Parada, B., et al., Omega-3 fatty acids inhibit tumor growth in a rat model of bladder cancer. Biomed Res Int, 2013. 2013: p. 368178.
- Beata, C., et al., Effect of alpha-casozepine (Zylkene) on anxiety in cats. Journal of Veterinary Behavior, 2007. 2(2): p. 40-46.
- Wallius, B.M. and A.E. Tidholm, Use of pentosan polysulphate in cats with idiopathic, non-obstructive lower urinary tract disease: a double-blind, randomised, placebo-controlled trial. J Feline Med Surg, 2009. 11(6): p. 409-12.
- Gunn-Moore DA, Shenoy CM. Oral glucosamine and the management of feline idiopathic cystitis. J Feline Med Surg. 2004 Aug;6(4):219-25. doi: 10.1016/j.jfms.2003.09.007. PMID: 15265477; PMCID: PMC10822600.
- Krause LR, Li E, Lilly ML, Byron J, Cooper E, Quimby J. Survey of veterinarians in the USA to evaluate trends in the treatment approach for non-obstructive feline idiopathic cystitis. J Feline Med Surg. 2024 Aug;26(8):1098612X241260716. doi: 10.1177/1098612X241260716. PMID: 39163476; PMCID: PMC11418619.
- Panchaphanpong, J., Asawakarn, T., Pusoonthornthum, R., Lulich, J.P. & Osborne, C.A. (2011) Plasma and urine glycosaminoglycan concentrations in cats with idiopathic cystitis and healthy cats and the effect of oral N‑acetyl‑D‑glucosamine supplementation. American Journal of Veterinary Research, 72(2), pp. 218–223.
- Panchaphanpong, J. (2009) Plasma and urine glycosaminoglycan concentrations in cats with idiopathic cystitis and the effect of oral N‑acetyl‑D‑glucosamine. Master’s thesis, Chulalongkorn University.

